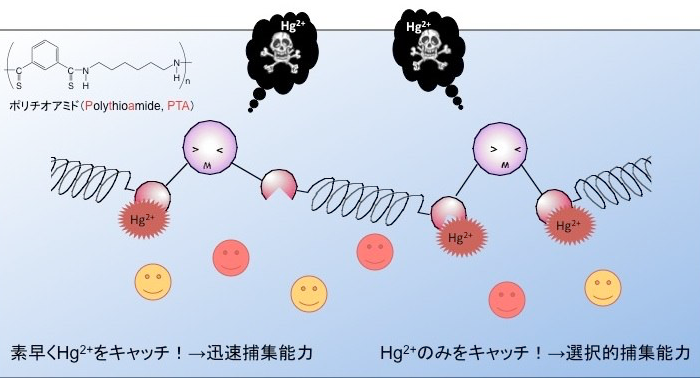
廃水中の水銀を選択的に吸着除去する
2010/04/01
The potential and feasibility of polythioamides as Hg(II) sorbents were evaluated. Powdered poly- thioamides quantitatively sorbed Hg(II) from an aqueous solution at pH 1–8. The sorption of Hg(II) on polythioamides obeyed the Langmuir adsorption isotherm; the sorption capacity was 0.70–0.85 g- Hg/g. Hg(II) was selectively separated from solutions containing 500 times larger amounts of Mn(II), Fe(III), Cu(II), Zn(II), and Pb(II) at pH 1. The tertiary polythioamide (PTA1) is soluble in chloroform and can be readily coated on a commercially available polymer resin, Amberlite XAD7HP. PTA1-coated resin as well as powdered PTA1 were applicable to the selective removal of Hg(II) from real wastewater.
Selective removal of mercury(II) from wastewater using polythioamides.
Shigehiro Kagaya, Hiroyuki Miyazaki, Masahiro Ito, Koji Tohda, Takaki Kanbara,
-
J.Hazard. Mater., 2010, 175(1-3), 1113-1115.
※ この研究は,筑波大 神原 先生と共同で行いました。